
Dipstick tests are "notoriously inaccurate, particularly in certain populations, such as the elderly," and culture-based methods have longer turnaround times, she added. The firm's focus is on UTIs because they are "neglected" diseases, Hayhurst said. It can also detect two or more pathogens in the same sample. coli, the sensitivity rises to about 93 percent, she said. The platform has been tested on hundreds of stored and fresh urine samples and can detect the infection-causing pathogens in about 85 percent of positive urine samples, with specificity around 90 percent. The automated reader indicates whether the result is positive or negative and what bacteria has been found in the sample. 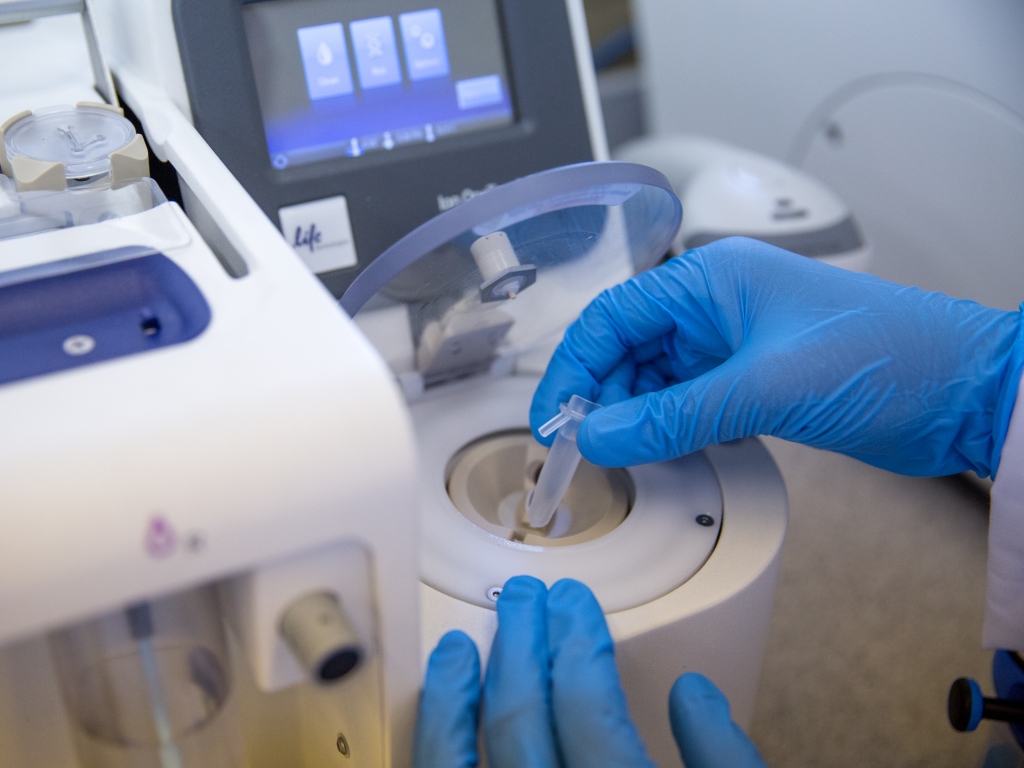
By using real-time LAMP, the test's result also doesn't depend on user interpretations of changes in color to determine a result, which can be a problem in some other tests that use endpoint LAMP, she said. The company used LAMP because it takes less time than PCR and can work with dirtier samples, so there's no need for sample purification, Hayhurst noted. The UTI panel will include assays for Escherichia coli, Klebsiella, Enterococcus, Proteus mirabilis, Pseudomonas aeruginosa, and Staphylococcus saprophyticus. Once the sample is added to the tubes, the tubes are put into the reader device, which takes readings every 10 seconds and converts the reaction data to a positive or negative result and determines which bacteria is causing the infection in about 40 minutes, she said. The sample is then deposited into eight separate pre-prepared tubes, six of which contain reagents for a different pathogen and two of which serve as positive and negative controls. To run the UTI test, a user dips the sampling device into a urine sample. The three-step workflow involves no sample processing or user interpretation, and the platform consists of a sample transfer device, tubes, and reader device, Hayhurst said. The firm's system is a portable, battery-powered molecular diagnostic instrument that relies on loop-mediated isothermal amplification (LAMP) to detect infectious disease-causing pathogens. In a presentation at the 33rd European Congress of Clinical Microbiology and Infectious Diseases this week, Llusern CEO Emma Hayhurst described the details of the Lodestar Dx platform and the company's plans for regulatory approval and commercial launch. COPENHAGEN – University of South Wales spinout Llusern Scientific is preparing its portable molecular diagnostic platform to launch in the next year, along with a panel for urinary tract infections.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
